22 Sabri and colleagues demonstrated that highly viscous multiple Pickering emulsions could be prepared with a single type of silica nanoparticle grafted with silanes and sodium alginate. 20,21 Contact angle hysteresis of microbowls can be leveraged to prepare multiple Pickering emulsions by using a silicone microbowl as a sole stabilizer. 19 They also prepared hierarchical porous polymeric microspheres and capsule clusters based on a double Pickering emulsion with a polymerizable middle oil phase. 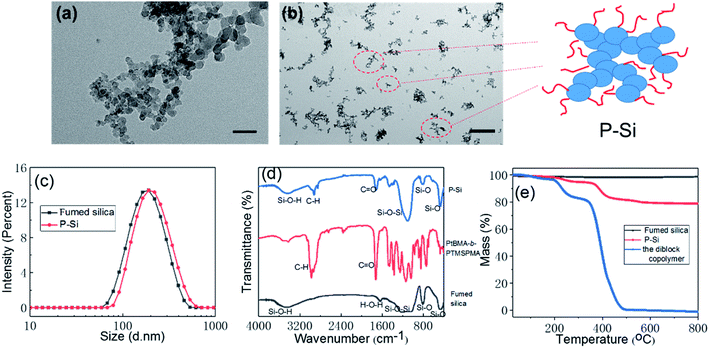
18 Ning and colleagues produced water-in-oil-in-water (W/O/W) double Pickering emulsions with hydrophobic silica H30 and hydrophilic mesoporous silica nanoparticles. 16,17 Fujii and colleagues demonstrated that the wettability of hydroxyapatite (HAp) particles could be tuned through the adsorption of poly( L-lactic acid) (PLLA) and that a stable multiple emulsion could be prepared using a combination of PLLA-coated HAp and pristine HAp particles. 15 Armes and colleagues tuned the wettability of silica particles by coating them with poly(ethylene imine) and then enabling a subsequent reaction with 1-undecanal through Schiff base chemistry. 14 They also found that multiple Pickering emulsions could be prepared from a mixture of hydrophilic and hydrophobic pigments. 
1,12,13 Binks and colleagues prepared stable multiple emulsions containing ionic liquids stabilized by silica nanoparticles that differed in their SiOH content. In general, multiple Pickering emulsions, also called “emulsions of emulsions,” have been prepared through a two-step emulsification process using a binary mixture of particles differing in wettability. 10 The wettability of particles can be tailored by modifying the surface chemistry and topology ( e.g., roughness). Particles that are completely wetted by water or oil are dispersed in this phase and cannot form an emulsion. 9 Hydrophilic particles with contact angle 90° are more suitable for the stabilization of water-in-oil (W/O) emulsions. Generally, the main wetting liquid forms the continuous phase and the other liquid forms the dispersed phase. 7,8 The type and stability of a Pickering emulsion are closely related to the wettability of the colloidal particles. 1–3 The inherent advantages of Pickering emulsions-their excellent stability and surfactant-free characteristics-make them attractive for numerous applications relating to food, 4 pharmaceuticals, 5 crude oil recovery, 6 and catalysis. Introduction Particle-stabilized emulsions, also known as Pickering emulsions, which have been largely ignored since they were first reported in early 1907, are recently receiving increasing academic attention. Multiple emulsion microreactors increase the area of the oil–water interface and are therefore more efficient than the commonly used O/W and W/O emulsion systems. Moreover, we demonstrated that multiple emulsion systems with adjustable pH are suitable for the development of an efficient and recyclable interfacial catalytic system. Using these SSMs as the sole emulsifier, we adjusted the pH to successfully produce both water-in-oil-in-water (W/O/W) and oil-in-water-in-oil (O/W/O) multiple emulsions through a single-step emulsification process. In this study, spherical SSMs were prepared via polymerization-induced self-assembly co-mediated with a binary mixture of macromolecular chain transfer agents: pH-responsive poly(2-(dimethylamino) ethyl methacrylate) and hydrophobic polydimethylsiloxane. Surface-segregated micelles (SSMs) with adaptive wettability have considerable potential for application in Pickering emulsions and bioanalytical technology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
